- Home
- About ANT
-
Products
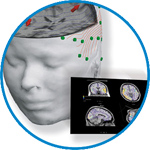
asa
asa is a highly flexible EEG/ERP and MEG analysis package with a variety of source reconstruction, signal analysis and MRI processing features.
.jpg)
eego mylab
The new frontier in multimodal brain research. With up to 16 kHz sampling rate, 256 EEG channels and unique software features, eego mylab gives you an unprecedented in-depth understanding of the human brain.

eego sports
eego sports offers complete freedom to collect high-density EEG data, bipolar EMG signals, and a variety of physiological sensor data, wherever and whenever required, with publish quality data in less than 15 minutes!

waveguard net
The waveguard net sets a new standard for research applications requiring high-density EEG data acquisition with quick preparation time, high flexibility, and subject comfort.
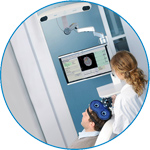
visor2
Our new and upgraded visor2 solutions integrate all the latest technologies for navigated rTMS, dual-coil navigation support, EEG-TMS recordings and pre-surgical evaluation for the highest quality in research and clinical procedures.
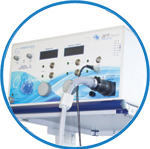
powerMAG ANT
The PowerMAG ANT 100 rTMS stimulator is designed for the specific needs of high-end TMS applications. Powerful high-frequency TMS as well as high precise single pulse and repetitive pulse protocols are combined in one single device.

xensor
xensor offers the solution for digitization of 3D electrode positions. xensor takes care of the whole procedure; it records, visualizes and stores positions acquired with a dedicated digitizer.

waveguard original
waveguard original is the cap solution for EEG measurements compatible with fMRI, MEG and TMS system. Use of active shielding guarantees performance in even the most demanding environments.
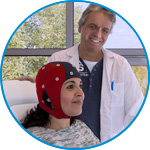
waveguard connect
waveguard connect EEG caps are a perfect match for hospitals and institutes aiming at reliable EEG, maximum uptime and great patient comfort! For optimal signal quality, the electrodes are made of pure, solid tin.

waveguard touch
waveguard touch is a dry electrode EEG cap. The unique Ag/AgCl coated soft polymer electrodes provide stable, research-grade EEG signals while maintaining subject comfort. The combination of these innovative dry electrodes and the industry-leading waveguard cap makes waveguard touch the best solution for dry EEG.
smartmove
smartmove allows planning of a complete TMS session ahead by defining stimulation sites based on anatomical MRI information and functional information like fMRI, PET or EEG/MEG.
Stay - References
- Support
- Events
- News
- Contact Us
You are here
eego™mylab
eego™mylab
Deeper insights through better technology.
The new frontier in multimodal brain research. With up to 16 kHz sampling rate, 32-256 EEG channels and unique software features, eego mylab gives you an unprecedented in-depth understanding of the human brain.
The battery powered system is available in 4 versions for recordings from 32 to 256 EEG channels. The cascaded 128- and 256-channel versions offer users a unique benefit; the high-density EEG stations can be split into separate (mobile) 64-channel EEG systems for individual use or group studies.
eego mylab is ideal for investigating all kinds of EEG paradigms, such as (SS)VEP, AEP, MMN, P300 and N2(pc) as well as for complex paradigms in combination with TMS, EMG or customized solutions such as MEG and tDCS/tACS. Thanks to the high sampling rate, high density and the unique active shielding technology, the quality of EEG remains optimal even in adverse environments.
In addition, the system’s functionality can be easily extended with EOG, ECG, EMG, real-time data access and physiological sensors for respiration, temperature, skin conductance and acceleration.
eego mylab applications >>
Core features and benefits
- Plug & play system with quick set-up times.
- Compact and light-weight system for use in clinical as well as research settings.
- Ultimate flexibility with interchanging amplifiers for use as cascaded and stationary units or as individual and mobile* systems.
- The system is available in 4 versions, for recordings from 32 to 256 EEG channels.
Available system configurations >> - Compatible with dry and gel-based EEG caps.
- Includes an input for recordings of up to 24 bipolar channels or a combination of EMG channels with a variety of physiological sensors for rich data of body and mind.
- High temporal resolution data thanks to one of the highest sampling rates in the industry: sampling rate of up to 16 kHz across all referential channels.
Amplifier specifications >> - Ultra-high signal range and programmable gain.
- Includes a wealth of software features designed for unconstrained and intuitive performance, ranging from practical recording workflow and subject management to advanced sensor value display and synchronized video recording. Software features >>
- Full integration of a wide range of external triggers.
- Excellent signal quality even in adverse environments due to the active shielding technology. more>>
- SDK for direct real-time data access (e.g. BCI, Neurofeedback).
- Free support and software updates (initial 6 months).
The eego™ amplifiers are CE marked medical devices, and have FDA clearance under 510(k).
* Purchase of additional items is required.

 Read more
Read more.jpg)




